In addition to Soberana Plus, one of the Cuban fortifiers, Soberana 02, also arrived on the Old Continent.
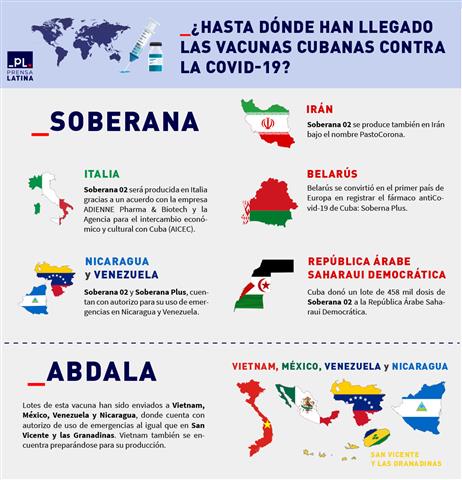
Soberana 02, designed at the Finlay Vaccine Institute (IFV) on the Caribbean island, as the Soberana Plus; They have been given the green light since April to be partially produced in Italy thanks to an agreement between the aforementioned entity, ADIENNE Pharma & Biotech and the Agency for Economic and Cultural Exchange with Cuba (AICEC) – both from Italy -.
https://www.youtube.com/watch?v=YgZgI0EoGoc
The memorandum signed during the BioHabana 2022 conference will allow the formulation, filling and packaging of the islet immunogen in the European country.
On this occasion, the Director General of IFV, Vicente Ferez, indicated that the agreement is closed due to the consideration of a possible moment for the entry of this vaccine in Europe or even in North America, noting that “this requires alliances to share the science.”
Ferrez also explained that the intention is, in the second phase, to be able to assess whether it can be fully produced in this Italian company Soberana 02 and other IFV vaccines.
With Italy, the threads connecting the anti-COVID-19 IFV products are even stronger. In November 2021, about 30 people between the ages of 19 and 59 from that country traveled to Cuba to volunteer for the Soberana Plus Turin clinical study.
The aim of the trial was to assess immunity in COVID-19 convalescents and people who had no history of the disease, but who had been immunized with other vaccines.
As a background to this collaborative analysis, at the “Amadeo di Savoia” hospital in Turin, sera from Cuban volunteers who had been vaccinated with Soberana Plus since July last year were evaluated.
The results demonstrated the ability of Soberana Plus to stimulate neutralizing antibodies against the alpha, beta and delta variants of the SARS-CoV-2 coronavirus, which causes the disease.
First in the world, Soberana Plus is specifically designed to reactivate immunity previously induced by other COVID-19 vaccines or by natural infection, Soberana Plus in Cuba was also used as a booster dose in the COVID-19 immunization plan, designed by IFV for two doses From her other product Soberana 02.
The combination of both has been shown in clinical trials to be 92.4 percent effective against symptomatic disease, according to the Soberana 02 Phase III study final results report.
This was the scheme used to immunize against COVID-19 for children aged 2-18 in the country, the first campaign in the world to reach more than 1.7 million infants.
The Cuban authorities described the childhood vaccination as a “complete success”. Well, about 70,000 cases have been averted, and since 2021 no child has died from COVID-19 in Cuba.
Not only in Europe is the “sovereigns” known. Soberana 02 arrived in Iran at the beginning of 2021 when it was still a vaccine candidate, with the aim of completing Phase III clinical trials.
“As part of cooperation with other countries in developing vaccines against COVID-19, 100,000 doses of Soberana02 have been sent to Iran’s Pasteur Institute, which will be used in clinical trials in that country,” the BioCubaFarma business group announced.
By the end of June of that year, Soberana 02 received a permit for emergency use in the Islamic Republic of Iran. The authorization was given to Iran’s Pasteur Institute (IPI), which will market the vaccine in Iranian territory, within the framework of a cooperation agreement signed with the Finlay Vaccine Institute.
In this way, the Persian country became the first in the world to produce the Cuban vaccines against COVID-19.
In that country, a center for serial production of the PastoCorona vaccine was opened, which became the result of the transfer of technology to the Cuban Soberana 02 vaccine to the Pasteur Institute.
On this side of the world, in Nicaragua and Venezuela; Both Sovereign 02 and Sovereign Plus have emergency use authorization.
Soberana 02 is among the vaccines approved by Venezuelan health authorities for use in children over the age of two, and in early 2022, a shipment of one million doses of Soberana Plus arrived in the Bolivarian country.
Health authorities have stated that they will use this latter drug to provide protection against reinfection by the Omicron variant.
For its part, the Nicaraguan Regulatory Authority has indicated since October 2021 that Soberana 02 is being introduced as a safe access therapeutic tool to reduce the transmission of Covid-19; Especially in children between the ages of 2 and 17 years.
Soberana 02 has also reached the arms of children in the Sahrawi Arab Democratic Republic. In February, Cuba donated a batch of 458,000 doses of this vaccine to this country for children’s use.
Abdullah also protects outside Cuba
Although the fortifications of the Sovereign Line are already known in many nations; The same is happening with Abdallah, a vaccine designed at the Cuban Center for Genetic Engineering and Biotechnology and the first vaccine in this nation and in Latin America to be manufactured against the SARS-Cov-2 virus.
Millions of doses of this blocker reached Vietnam, Mexico, Venezuela and Nicaragua, and when applied in a scheme of three doses every 14 days, it showed an efficacy of 92.28% against symptomatic disease. Furthermore, it has shown 100 percent efficacy in preventing severe systemic illness and death from COVID-19.
In September 2021, at the request of the Center for Research and Production of Vaccines and Biomedical Preparations, the Ministry of Health of Vietnam approved the import and use of the Al-Abdullah vaccine in this Indochina country.
In the same month, the first batch of the Cuban vaccine arrived in that country, consisting of 900,000 doses purchased by Vietnam and another 150,000 doses donated by the Caribbean country.
During the reception of that first shipment, Vice Foreign Minister Dang Huangjiang thanked Cuba for the doses, out of a total of 10 million agreed with the largest of the Antilles, and from where also the vaccine production technology in Cuba would be. Transferred to Vietnam this country.
In Nicaragua, Abdallah has been approved for emergency use since October 2021; Venezuela signed a contract to supply 12 million units of immunity with the Caribbean island in the same year.
At the end of 2021, the Federal Commission for the Protection of Health Risks (Cofepris) of the Government of Mexico ruled in authorizing the emergency use of Abdallah.
As a reference national regulatory authority, qualified by the Pan American Health Organization; Cofepris decisions are recognized by many countries in the region, so it is likely that approved vaccines will be used in other countries.
Being a member of the International Conference on Coordination of Technical Requirements for the Registration of Pharmaceutical Products for Human Use, all decisions of this authority are made based on the technical and scientific evidence presented.
In December of the same year, the Ministry of Health of Saint Vincent and the Grenadines announced on its Facebook social network account that Abdullah was available at vaccination sites in that country.
With this announcement, that country became the first in the Caribbean Community (Caricom) to allow one of the Cuban vaccines against Covid-19.
And who certified?
Despite all the evidence presented above, many skeptics will say that Cuban vaccines still lack recognition from the World Health Organization (WHO); And they are not without reason. Just make it clear; Although the problem has persisted longer than expected, each country can authorize the use of a vaccine whether or not it has been approved by that international entity.
But this does not mean that Cuba is stalled in the process.
This was recently confirmed by the Director-General of the World Health Organization, Tedros Adhanom, who confirmed last May that the agency is following the process of certification of vaccines against Covid-19 created and developed in Cuba.
Since March, the Caribbean Pharmaceutical and Biotechnology Industry Group (BioCubaFarma) has detailed on its Twitter account that the World Health Organization has been informed of the completed document to be reviewed by experts and start the process of international recognition of Abdullah’s vaccine.
The entity in the same social network indicated that the Center for Genetic Engineering and Biotechnology (CIGB) responsible for this immunization began the official exchange with the health entity that month.
In mid-February this year, Director of BioCubaFarma, Eduardo Martinez, explained that the company was working on the text, consisting of several chapters with findings on clinical and pre-clinical research, pharmaceutical development, as well as everything related to the facilities produced, an aspect that has been modified .
Regarding the operations followed with Abdullah for this assessment, Martinez noted that the decision was made to change the production site to the recently opened Mariel plant, located in that industrial pole, west of Havana.
The CIGB-Mariel-Tech-Industrial Park, operating since November 2021, is the newest in Cuba and one of the most advanced in Latin America and the Caribbean with quality control laboratories and warehouses for raw materials and finished products.
It also has botanicals for active ingredient vaccines and complete immunity in liquid, powder, and spray formulations.
Martinez stressed that the goal is for the representatives of the World Health Organization to visit the production factories of this entity, where they will conduct the necessary inspection to obtain the permit later and include it in the list of products recognized by the United Nations.
“Cuba always maintains exchanges with the WHO/PAHO representation on everything related to prequalification for immunity against the Covid-19 virus,” he said.
And the statement is no less true. Since September last year, the representative of the Organization of American States and the World Health Organization (PAHO / WHO) on the Caribbean island, Jose Moya, informed the Associated Press of a virtual meeting between specialists from Havana. Geneva and Washington to exchange information, coordinate documents, and set timetables.
Cuba’s solidarity not only filled the world with white coats; It is also arriving in several countries in lamps that can save millions of lives. The vaccines developed on this island bear the commitment, effort and capacity of scientists who have more than three decades of experience producing their own immunogens.
(Taken from PL)

“Unapologetic tv specialist. Hardcore zombie trailblazer. Infuriatingly humble problem solver.”
